 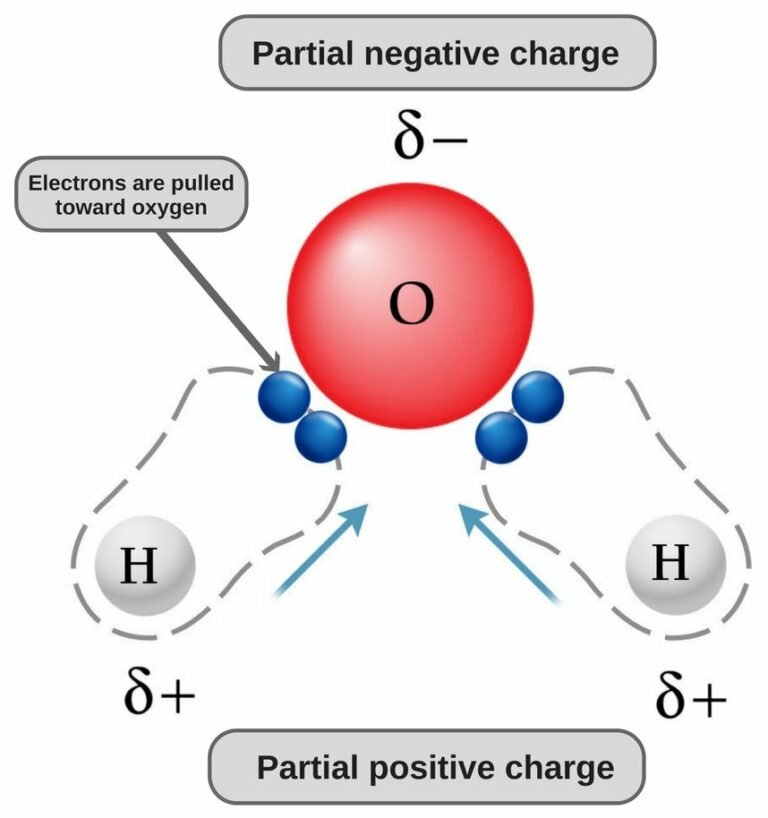
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. We designate SF 4 as AX 4E it has a total of five electron pairs. Versions 12 and higher have bromine-containing molecules.\).ģ. In this tutorial, we will discuss H2CO lewis structure, molecular geometry, polar or nonpolar, bond angle, hybridization, etc. It is the simplest form of aldehyde compromised of one oxygen, one carbon, and two hydrogen atom. Versions 8 and higher have a few substituted benzenes with more than six heavy atoms. It is an organic compound with the chemical formula H2CO. The bent shape of the molecules leads to gaps in the hydrogen bonding network of ice. A: We need a localized electron model to describe the bonding in H2O. 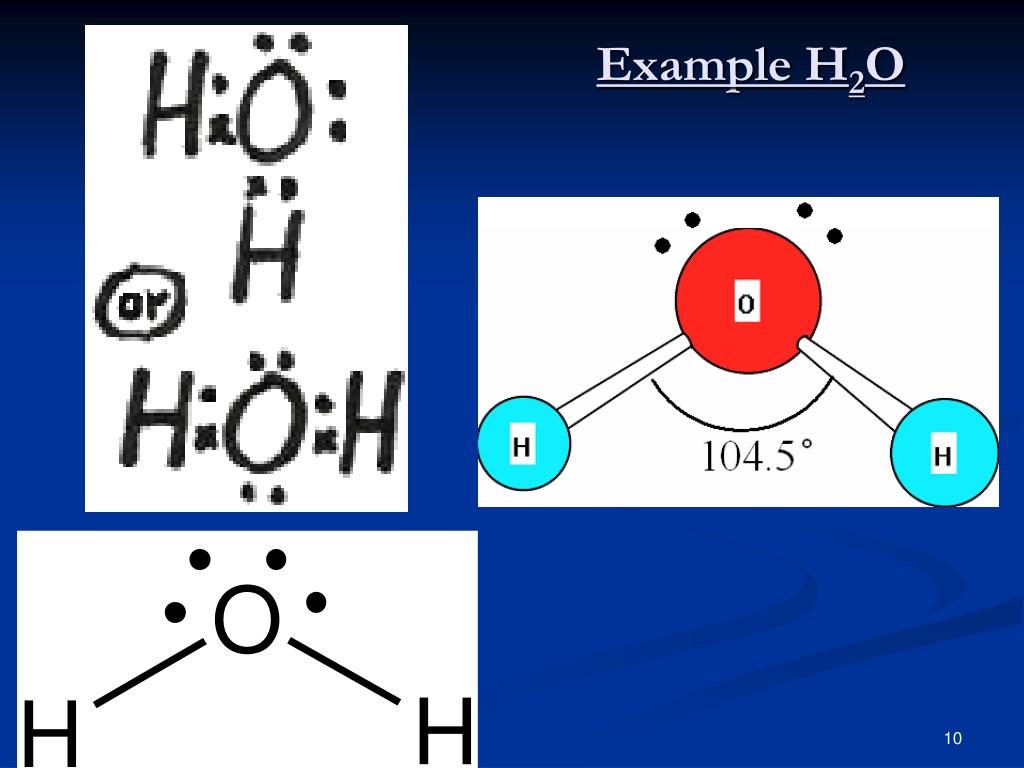
In NF3, the central nitrogen atom has four electron groups surrounding it: three single bonds( three bonded pairs) and one lone pair. We can also predict electron geometry via electron groups through VSEPR theory. Each oxygen atom has an approximately tetrahedral geometry that includes two covalent bonds and two hydrogen bonds. A: Movements of electrons are only possible when there are lone pair of electrons or pi-bonds and the Q: Use the localized electron model to describe the bonding in H2O. According to the VSEPR chart, the molecular geometry of nitrogen trifluoride is trigonal bipyramidal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
